1. INTRODUCTION
Water is a highly important resource for humans and other living organisms. It is used in households, for industrial and agricultural purposes, and for recreational activities such as fishing (Alkhatib et al., 2015; Yazid et al., 2021). Ensuring the quality of well water for consumption is essential for community development, as water may be contaminated and must undergo proper treatment before being supplied to households as tap water (Yang et al., 2019). High iron (Fe) concentrations can cause health problems related to mutations in genes responsible for absorbing iron and cause hemochromatosis, which can lead to liver, heart, and pancreas damage. Furthermore, it could be a precursor for diseases related to the heart and central nervous system, liver cirrhosis, diabetes, and nausea (Das et al., 2023).
Drinking water quality guidelines and standards established by the Indonesian Government are designed to ensure clean, safe, and high quality water for human consumption and to protect public health. According to the Minister of Health Regulation of Indonesia No. 2/2023, clean water must meet the established physical, chemical, and biological quality parameters to ensure it is safe for human consumption (Ministry of Health, 2023). These water quality requirements are in accordance with the World Health Organization (WHO) guidelines for drinking-water quality (WHO, 2022).
In reality, in the Special Province of Yogjakarta, Indonesia, water quality varies, and has particularly high Fe and Mn concentrations, which is a critical problem; for example, in Pelemsewu, Bantul District, Special Province of Yogyakarta, the water has high levels of Fe and Mn and appears highly turbid, but it is still used for daily needs such as drinking, bathing, and washing. The high Fe concentration in the water leaves reddish-orange stains on bathroom fixtures, such as bathtubs, showers, and faucets, which are difficult to remove, and causes a decline in the color quality of washed clothes; high Fe concentrations cause clothes to turn red, yellow, or brown, making them appear dirty and unhygienic (Chaturvedi et al., 2014; Das et al., 2023). However, adsorption using activated carbon—an approach that has been used for decades and proven effective in removing heavy metals from water—can be applied to address this problem (Saeed et al., 2020). Activated carbon adsorbs substances because of its high porosity, large surface area, and small particle size (Dungani et al., 2022; Singh et al., 2008; Sutapa et al., 2024b). Activated carbon is an appropriate filter material for hazardous materials and water and can be used to improve the quality of drinking water and wastewater (Schröder et al., 2007).
The production of bamboo chopsticks produces solid waste that cannot be utilized for the chopsticks. This waste is highly abundant and is generally not utilized optimally (Chen et al., 2017). Studies have been conducted on the conversion of bamboo waste into solid biofuel as an alternative to burning and co-firing with other resources (Chen et al., 2017). However, bamboo waste can also be used as a raw material to produce activated carbon (Choy et al., 2005; Mahanim et al., 2011; Zhang et al., 2014). Activated carbon is a suitable adsorbent for removing trace contaminants from air, soil, and water because of its high adsorption capacity (Skoczko and Guminski, 2024). The use of Ampel bamboo waste from the chopsticks industry to produce activated carbon is an innovative and practical approach, given the sustainability and biological availability of bamboo as a renewable resource; bamboo is a fast-growing plant (ranging from 3–5 years) with a high level of production, making it a major global biomass resource (Jeon et al., 2018; Sumardi et al., 2022). Furthermore, bamboo has been cultivated in Indonesia in forests and with edible crops in residential gardens (Park et al., 2018).
In this study, charcoal was produced from Ampel bamboo waste as an eco-friendly material (Hwang and Oh, 2022). The charcoal was then converted to activated carbon and used to filter well water from the rural communities of Pelemsewu to improve its quality. The aim of this study was to determine the optimal dose and contact time of Ampel bamboo waste-derived activated carbon for improving well water quality. Specifically, we evaluated its effectiveness in removing Fe and Mn to ensure safe and high-quality water for human consumption.
2. MATERIALS and METHODS
Well water samples were obtained from Pelemsewu, Bantul District, Special Province, Yogyakarta, Indonesia. Ampel bamboo waste was obtained from the CV Jaya Abadi chopstick manufacturing factory in Tasikmalaya, West Java, Indonesia. This study was conducted using a completely randomized design with two treatment factors: activated carbon dose (0.5, 1.25, and 2 g/L) and contact time (15, 45, and 75 min) with three replicates. Analysis of variance (ANOVA) was performed, followed by Tukey’s honest significant difference test to test for significance analysis.
The bamboo waste was cut into smaller rectangular pieces (2 × 7 cm dimensions) and air-dried until the moisture content reached 10%–15%. The air-dried bamboo was carbonized at a temperature of 400°C for ± 3 h using an electric retort. The carbonized bamboo (charcoal) was ground into powder form and filtered through a 10-mesh sieve, retaining the particles on a 20-mesh sieve to ensure a homogeneous particle sizes (Fig. 1).

Carbon Activation was conducted as follows: 52.61 g of bamboo charcoal was inserted in a 200 mL curable porcelain cup and heated at 900°C for 90 min in a furnace (FB 1410M-33; single setpoint with a capacity of 2.1 L; power consumption, 1,520 W; temperature range, 100°C–1,100°C; temperature stability, ± 5.0 at 1,000°C; and electrical requirements of 240 V 50/60 Hz). This process was repeated for five replicates. The characteristics of the resulting activated carbon were evaluated and subsequently used in batch experiments to remove Fe and Mn from well water and improve the quality.
Morphological changes in the charcoal surface before (batch 1) and after (batch 2) activation were investigated using a Tabletop scanning electron microscope (SEM) TM4000Plus (Hitachi High-Technologies, Tokyo, Japan) at an acceleration voltage of 10 kV. This instrument allowed the analysis of samples without the need for conductive coating.
X-ray diffraction (XRD) analysis of charcoal and activated charcoal was performed using a Bruker D2 Phaser X-ray Diffractometer (Bruker, Berlin, Germany) operating at 30 kV and 10 mA, equipped with Cu Kα radiation (λ = 1.54184 Å). Diffraction patterns were collected over a 2θ range of 5°–80°.
A total of 1,000 mL of well water was mixed with Ampel bamboo waste-derived activated carbon at doses of 0.9, 1.2, and 1.5 g/L, and contact times of 15, 45, and 75 min, with three replicates of each combination. The resulting solution was mixed and stirred in an Erlenmeyer flask using a magnetic stirrer (C_MAG HS 7, IKA, Shanghai, China) at a temperature of 30°C and a speed of 1,000 rpm, and then filtered through filter paper, following the method described by Subroto et al. (2025), with slight modifications. This method used the following variables: dose ranging from 0.2–0.7 g/L, contact time ranging from 30–150 min, and stirring speed between 100 and 400 rpm.
The filtrate was then analyzed for quality parameters, including Fe, Mn, color, pH, turbidity, and water hardness. The treated well water was compared with the Indonesian drinking water quality standards according to the Minister of Health Regulation No. 2/2023.
The parameters and standards used to evaluate well water quality after treatment with activated carbon include Fe and Mn concentrations (SNI 6989.04-2019), color (SNI 6989.80-2011; Badan Standardisasi Nasional, 2011), pH (SNI 6989.11-2019; Badan Standardisasi Nasional, 2019a), turbidity (SNI 06-6989.25-2005; Badan Standardisasi Nasional, 2005), and total hardness (SNI 06-6989.12-2004; Badan Standardisasi Nasional, 2004).
Fe and Mn measurements were performed using an atomic absorption spectrophotometer (SSA Hitachi Z-2000, 240 V Model 7 JO-8024) and following the potentiometric method (SNI 6989-04-2019; Badan Standardisasi Nasional, 2019b). The following equation was used to determine Fe and Mn concentrations:
where C is the metal concentrations (mg/L) obtained from measurement results and Fp is the dilution factor.
Color content was evaluated using a spectrophotometer at wavelengths ranging from 450–465 nm using a standard Pt-Co solution to measure the true color of the test sample solution based on Beer’s law.
The following equation was used to determine color content:
where D is the value obtained from the calibration curve (expressed in Pt-Co units) and Fp is the dilution factor.
pH was measured based on potentiometric hydrogen ion (H+) activity using a pH meter LUTRON PH-207. Briefly, the electrode was rinsed with mineral-free water and dried with soft tissue paper. The electrode was then submerged in the test sample until the pH meter provided a correct and stable reading. The readings were then recorded. Temperature was measured simultaneously with the pH measurement, and the results were recorded in a laboratory worksheet. Finally, the electrode was rinsed with mineral-free water again.
A nephelometer was used to measure water turbidity. The amount of light that was absorbed and refracted in the test sample was compared with that of a standard (clear) suspension.
The following equation was used to calculate turbidity:
where T is the turbidity (NTU) of the diluted sample and Fp is the dilution factor.
The total hardness of the water and wastewater was determined using the EDTA titrimetric method, with a lower limit of 5 mg/L. This method was used for the colorless water samples.
The following equation was used to test water hardness:
where VC.u is the volume of test sample solution (mL), VEDTA(a) is the average volume of Na2EDTA standard solution for total hardness titration (mL), and MEDTA is the molarity of Na2EDTA standard solution for titration (mmol/mL).
3. RESULTS and DISCUSSION
Fig. 2 shows a comparison of the surface morphologies of the charcoal and activated carbon. The SEM images show that activated carbon exhibits significantly higher porosity than ordinary charcoal. This enhanced porosity and removal of the inert material in the cell lumens of the activated carbon [Fig. 2(b)] indicate its large surface area, which is essential for its effectiveness in adsorption applications (Zhu et al., 2024).
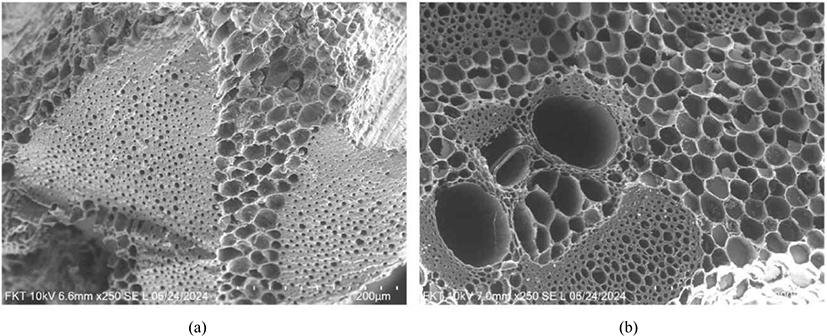
Analysis of the SEM cross-sectional images showed that, after activation, the sclerenchyma cells and phloem were cleared of inert material, resulting in open pores and clearly defined cell walls (Fig. 2).
XRD analysis was conducted to investigate the crystalline structures of the bamboo charcoal and activated carbon (Lazzarini, 2017). The patterns generally revealed an amorphous structure for charcoal and activated carbon. However, the distinct peaks indicate the presence of crystalline characteristics (Fig. 3). Both materials exhibit distinct peaks at approximately 2θ = 23°, corresponding to a graphite-like microcrystalline structure (002) lattice plane. In contrast to charcoal, activated carbon displayed sharper peaks, indicating a greater regularity in its crystalline structure due to the higher-temperature treatment. Additionally, activated carbon shows a peak at approximately 2θ = 43°, which corresponds to the (100) lattice planes of a graphite-like microcrystalline structure (Wang et al., 2014). To further quantify the structural changes, the crystallinity index (CrI) was measured using the method described by Segal et al. (1959): CrI = [(I002 – I am) / I002] × 100%. The analysis revealed that CrI increased from 40.49% in bamboo charcoal to 51.06% in the activated carbon.
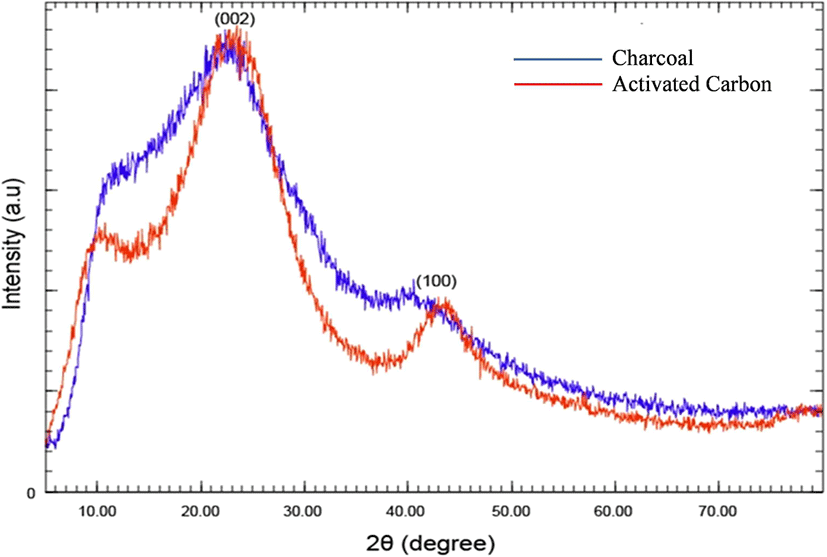
Based on the peak intensity, this XRD pattern was highly similar to the XRD pattern of the activated carbon of palm kernel shells collected from the palm oil industry in Muara Enim (Damayanti et al., 2025). Furthermore, the XRD pattern of activated carbon from bamboo waste was similar to that of bamboo carbon activated using steam-based methods (Zhu et al., 2024). These findings imply that before heat activation, the charcoal exhibited an amorphous structure, and subsequent thermal treatment significantly improved its graphitic characteristics (Wang et al., 2018).
Finally, the interlayer spacing (d002) was checked using Bragg’s equation [d002 = λ / (2 sin θ)]; Almugren et al., 2022). The analysis shows that the spacing decreased from 0.3897 nm for charcoal to 0.3798 nm for activated carbon. This indicates that the activation process allowed the graphite sheet to pack more tightly.
This study used the most effective method to produce activated carbon from Ampel bamboo waste, which was described by Sutapa and Ramba (2021). The activated carbon originated from the activation of Ampel bamboo waste charcoal at a temperature of 900°C for 90 min. Table 1 shows the quality parameters of the activated carbon from Ampel bamboo waste, that is, moisture content, volatile matter, and fixed carbon content, compared with the Indonesian quality standard SNI 06-3730-1995.
Data from Sutapa and Ramba (2021).
Moisture content refers to the amount of water physically bound to the activated carbon under normal conditions. In contrast, carbon ash refers to the residue that remains after the material is burned. Ash contains inorganic material that is bound to the activated carbon, and generally ranges from 2%–10%. The lower the ash content, the better the activated carbon, as high ash content can lead to increased hydrophilicity and catalytic effects, resulting in a restructuring process during the regeneration of used activated carbon (Nurul’ain, 2007). High levels of volatile matter indicate that the surface of the activated carbon contains non-carbon compounds and substances, which originate from the interaction of carbon with water vapor; therefore, a low volatile matter content implies high porosity of the adsorbent because less volatile matter remains clogged in the pores (Haji et al., 2013; Qian et al., 2007). This lower volatile matter content is caused by the removal of volatile compounds during carbonization at high temperatures (Ju et al., 2020). The fixed activated carbon content in this study was 72.41%, which complied with the SNI 06-3730-1995 standard. A higher value of fixed carbon leads to better iodine absorption. These results align with those of a study conducted by Sutapa et al. (2024a) on activated carbon from the sapwood waste of fast-growing teak. They reported that the highest iodine adsorption value was found at high fixed-carbon values.
The characteristics of the adsorption capacity of Ampel bamboo waste-activated carbon include the adsorption capacities for benzene (%), iodine (mg/g), and methylene blue (mg/g). Its adsorption capacity for benzene was 8.32%. The capacity for benzene adsorption is related to the surface area of activated carbon; greater benzene absorption indicates a higher level of porosity in the activated carbon (Isinkaralar, 2022). The absorption capacity for benzene is an indicator of the quality of activated carbon in absorbing pollutant gases (Mohammad-Khan and Ansari, 2009), thus, the absorption capacity for benzene is not a relevant indicator for the removal of Fe and Mn.
The iodine adsorption capacity of the activated carbon was 752,818 mg/g, meeting the SNI 06-3730-1995 quality standard. According to Idrus et al. (2023), activated carbon with a high iodine adsorption capacity that meets quality standards can absorb adsorbates or dissolved substances. Based on the iodine adsorption capacity, the surface area of the activated carbon was calculated according to an equation described by Mianowski et al. (2007): SA = 0.986 × IN, where SA is the surface area and IN is the iodine absorption number. The calculated surface area of activated carbon was 742 m2/g. Thus, the high iodine adsorption capacity of activated carbon indicates a high capability to absorb Fe and Mn. Therefore, activated carbon from Ampel bamboo waste can be used as an alternative water filter media to improve water quality. As shown in Table 1, the methylene blue adsorption capacity was 97.90 mg/g, which does not meet the SNI 06-3730-1995 quality standard. The magnitude of methylene blue absorption capacity is indicative of the ability of activated carbon to absorb colored liquid material solutions with a molecular size of 15 Å or 1.5 nm (Alimah, 2017). Although the activated carbon did not meet the absorption standard with methylene blue, the produced Ample bamboo-activated carbon removed some color from the well water.
Tables 2 and 3 show the results of using activated carbon from Ampel bamboo waste to improve well water quality. After treatment, Fe and Mn concentrations were reduced. Furthermore, quality improvements were observed in terms of pH, color, turbidity, and hardness. This result showed the effectiveness of activated carbon as an absorbent for improving water quality. Fig. 4 shows the effect of the Ampel bamboo waste-activated carbon on the color of the Pelemsewu well water before and after treatment. The well water, which was initially cloudy, became clear after treatment.
| Source | DF | Iron (Fe) | Manganese (Mn) | Color | pH | Turbidity | Total hardness |
|---|---|---|---|---|---|---|---|
| Adsorbent doses (A) | 2 | NS | ** | NS | ** | ** | NS |
| Contact time (C) | 2 | NS | ** | NS | ** | NS | NS |
| A × C | 4 | NS | ** | NS | NS | NS | NS |
| Error | 18 | ||||||
| Total | 27 |

High Fe concentrations in groundwater can cause health problems in the skin and the digestive, respiratory, and nervous systems (Achary, 2014). High Fe concentrations in well water can affect its quality; for example, the water has a metallic taste, unpleasant odor, and discoloration (Subroto et al., 2025). Kumar and Sinha (2018) reported that in Moradabad City, India, the highest Fe concentrations in well water were recorded during the summer months, and slightly lower concentrations were recorded in winter. Water containing high Fe levels causes stains on clothing and sanitary equipment and gives a bitter astringent taste.
The Fe concentration in the Pelemsewu well water was 0.56 mg/L. After treatment with activated carbon, the average Fe concentration in the well water was 0.06 mg/L, ranging from 0.03–0.09 mg/L (Table 2). Based on the ANOVA, the Fe concentration in the well water after treatment was influenced by the interaction between the adsorbent dose and contact time; however, they showed no significant effect, but Fe levels in the well water were still reduced. Fig. 5 shows that treatment with an activated carbon adsorbent dose of 0.5 g with a contact time of 45 min decreased Fe concentration by 94.73% (from 0.56–0.03 mg/L). The Fe concentration in the well water after treatment with Ampel bamboo waste-activated carbon showed a very high reduction because of the small distance between Fe ions, making it easier for them to penetrate the pores of the activated carbon (bin Jusoh et al., 2005). The results showed that well water treatment using activated carbon from Ampel bamboo waste produced the lowest reduction in Fe concentration (80.33%). This result was affected by the higher iodine absorption capacity (752.82 mg/g). Furthermore, according to Dastgheib and Rockstraw (2002), activated carbon has a higher adsorption capacity for Fe because of its electronegativity, which is similar to its ability to attract electrons; higher electronegativity corresponds to a higher adsorption capability for metal ions. In this case, Fe, which has a positive charge, is attracted to a negative charge on the activated carbon surface.
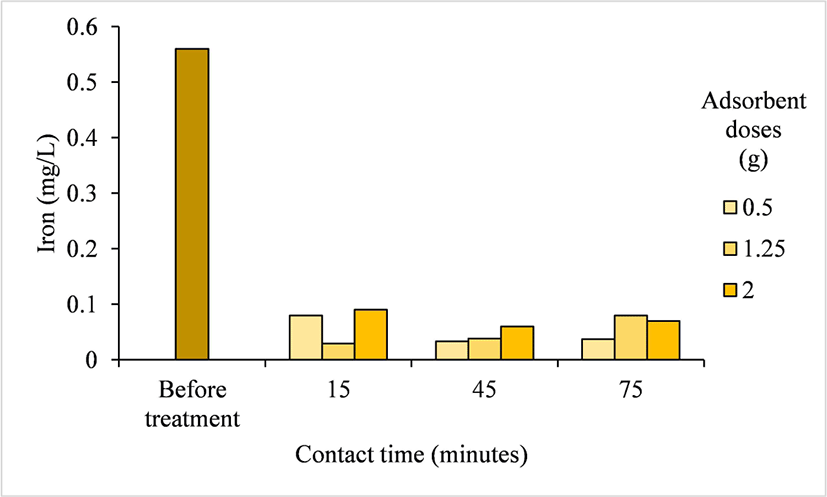
This value is slightly lower than that of coal-based activated carbon modified with NiFe2O4, which can reduce Fe concentration by 96.12% (Subroto et al., 2025). The results showed that well water treated with Ampel bamboo waste-derived activated carbon at doses of 0.5, 1.25, and 2 g/L and contact times of 15, 45, and 75 min met the Indonesian standards of the Minister of Health Regulation No. 2/2023 for Fe, with a maximum allowable concentration of 0.2 mg/L. These findings show Ampel bamboo waste-derived activated carbon is effective in improving the quality of water from the Pelemsewu wells. Therefore, treatment using activated carbon is a viable solution for improving the quality of water containing high Fe concentrations.
The ability of activated carbon to remove Mn from well water is an important characteristic because Mn pollution pose significant risks to public health. Mn at concentrations > 300 μg/L have been shown to have detrimental neurological effects on infants (Dvorak and Schuerman, 2021). The initial Mn concentration of the Pelemsewu well water was 0.81 mg/L, but after treatment, the average Mn concentration was reduced to 0.57 mg/L, ranging from 0.41–0.66 mg/L (Table 2). Based on the ANOVA results, the Mn concentration in the treated well water was significantly affected by the interaction between the adsorbent dose and contact time (p < 0.01). Fig. 6 shows that Mn concentration decreased by 49.72% with increasing adsorbent doses and contact times. The largest decrease occurred after treatment with 2 g/L activated carbon for 75 min. Badawi et al. (2017) stated that as the dose increases, the adsorption on the adsorbent surface area increases, thereby reducing the amount of pollutants. This result is similar to that of Subroto et al. (2025), who stated that both activated carbon and activated carbon/NiFe2O4 composites have similar affinities for reducing Mn concentrations. The mixing of activated carbon and NiFe2O4 improved the separation process and increased the adsorption capacity. Ampel bamboo waste-derived activated carbon has a porous structure that increases its absorption capacity for Mn ions (Budinova et al., 2009). This high absorption capacity for Mn is attributed to the positively charged Mn ions, which interact and bind to functional groups on the surface of the activated carbon through chemical bonding (bin Jusoh et al., 2005). Functional groups attract cations, promoting ion exchange. Although the results showed a reduction Mn concentration of Pelemsewu well water after treatment (0.81–0.57 mg/L), this concentration does not meet the Indonesian clean water quality standards of the Minister of Health Regulation of Indonesia No. 2/2023 for Mn, which is 0.1 mg/L.
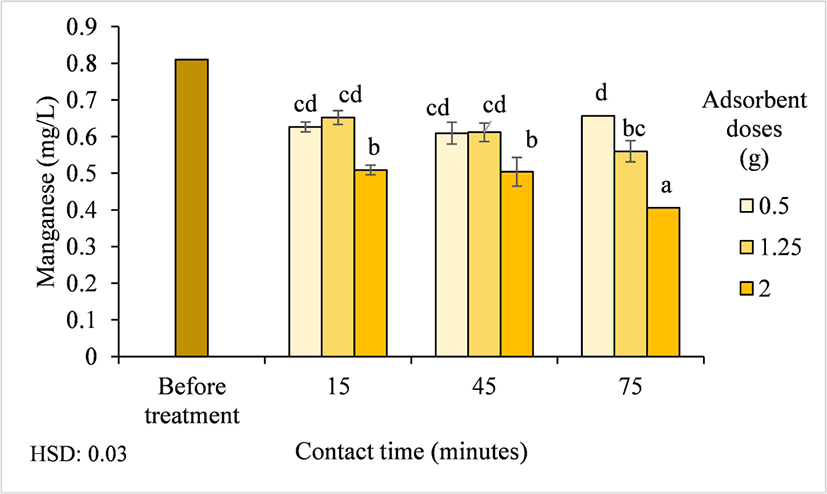
Another factor contributing to the different activated carbon adsorption capacities for Mn and Fe ions is their ionic radii. Fe has a smaller ionic radius than Mn; the smaller ionic radius of Fe allows them to easily penetrate the micropores of activated carbon.
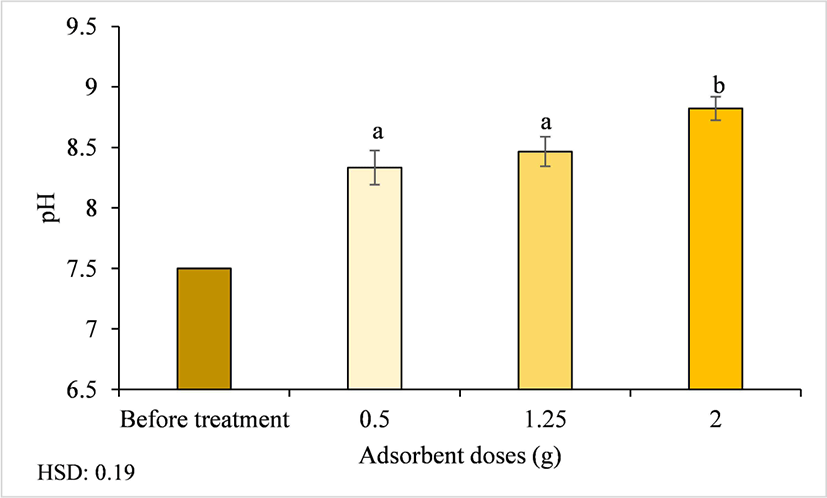
The pH can be used as an indicator of water hardness. Pure water has a pH of 7, surface water has a pH range of 6.5–8.5, and groundwater has a pH range of 6–8.5. The biological effects of low pH include to gill damage, mucus buildup on the gills, stunted growth, problems with ion regulation, and reproductive failure, which leads to a reduction in the number of aquatic species in the environment and replacement of acid-sensitive species with acid-tolerant species (Dirisu et al., 2016; WHO, 2022). The pH of water influences the rate of metal corrosion and the efficiency of disinfection. Thus, the pH of water entering the distribution system for households or industries must be maintained to minimize the corrosion of water channels and pipes. Corrosion and failure of water pipes can contaminate drinking water and affect its taste, odor, and appearance (WHO, 2022). Furthermore, the pH is an important factor influencing the adsorption of Fe (II) ions (Karim et al., 2024). The initial pH of the Pelemsewu well water was 7.5, and after treatment with activated carbon from Ampel bamboo waste the average pH increased to 8.5, ranging from 8.2–8.9 (Table 2). Based on the ANOVA results, the adsorbent doses significantly affected the pH (p < 0.01). Fig. 7 shows that the higher the dose of activated carbon, the higher the pH value of the water. The activated carbon influenced the pH by decomposing the metal elements in the well water into metal and hydroxide ions (OH–). Metal ions are attracted to the surface of activated carbon, which interacts and binds with acid groups so that H+ ions are reduced, and only OH– ions remain (Heriyani and Mugisidi, 2016). After treatment with Ampel bamboo waste activated carbon, the pH of Pelemsewu well water met the Indonesian drinking water quality standards (range of 6.5–8.5) of the Minister of Health Regulation No. 2/2023.
The color of water and wastewater is an important parameter of water quality as it affects photosynthesis by phototrophic organisms and influences dissolved oxygen content; thus, color changes can result in an ecological imbalance. In addition, some colored substances in water can be categorized as toxic or carcinogenic (Bao et al., 2017). The color content of the well water of Pelemsewu Bantul was initially 20 TCU. After treatment with Ampel bamboo waste activated carbon, the average color content of the well water was reduced to 1.78 TCU, ranging from 1–3 TCU (Table 2). The color content of the well water after treatment was influenced by the interaction between the activated carbon dose and contact time. The highest reduction in color content reached 95% (from 20–1 TCU), which occurred at adsorbent doses of 0.5 g and 1.25 g with a contact time of 15 min and at an adsorbent dose of 2 g/L with contact times of 15, 45, and 75 min (Fig. 8).
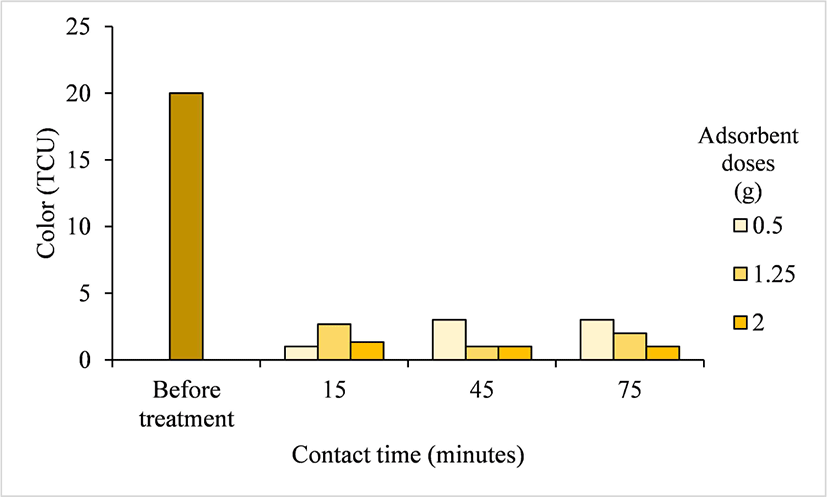
This considerable decrease in color content is correlated with a reduction in Fe concentration in the Pelemsewu well water after treatment because of the microporous structure of activated carbon, which absorbs chemical particles (Gürses et al., 2006). The color content of the well water after treatment met the Indonesian quality standards of the Minister of Health Regulation of No. 2/2023, which is 10 TCU. This suggests that Ampel bamboo waste-derived activated carbon is a promising water purification medium.
The higher the number of suspended solids in the water, the more turbid the water. Turbidity is a parameter used worldwide to describe the quality of drinking water (WHO, 2022). In this study, the initial turbidity of well water from Pelemsewu was 100 NTU. After treatment with activated carbon from Ampel bamboo waste, the turbidity was reduced to 0.79 NTU, ranging from 0.43–1.13 NTU (Table 2), which is a significant reduction (> 99%). The reduction in turbidity was 99.47%, 99.26%, and 98.91% at adsorbent doses of 0.5, 1.25, and 2 g/L, respectively. This result is consistent with that of Subroto et al. (2025), who proved that coal-based activated carbon modified with NiFe2O4 can reduce the turbidity of water. The reduction in turbidity observed in this study is greater than that reported by Lin et al. (2017), who found a 22.59% decrease using multi-layer water filtration at a flow velocity of ± 5 mL/min through Moso bamboo-derived activated carbon. This is likely due to the higher iodine absorption capacity of Ampel bamboo waste-derived activated carbon, which has a well-developed microporous structure that enhances its ability to remove suspended substances from the water (Budiman et al., 2019).
The turbidity of well water treated with Ampel bamboo waste-derived activated carbon at doses of 0.5, 1.25, and 2 g/L and contact times of 15, 45, and 75 min, respectively, met the Indonesian quality standards of the Minister of Health Regulation No. 2/2023, which is a maximum turbidity of < 3 NTU.
Hard water is harmful to human health and can cause diseases. Over the last five decades, the number of deaths related to cardiovascular disease due to the hardness of drinking water has increased (Akram and Rehman, 2018; Sengupta, 2013). The initial value of the total hardness of the Pelemsewu well water (202.99 mg/L) was significantly reduced after treatment with Ampel bamboo waste-derived activated carbon. The results showed that the average total hardness concentration of Pelemsewu well water after treatment was reduced to 185.44 mg/L, ranging from 165.15–197.02 mg/L (Table 2). The highest reduction in total hardness concentration (16.67%) occurred when the water was treated with an adsorbent dose of 1.25 g for 75 min (Fig. 9). This result is lower than that reported by Lin et al. (2017), who used Moso bamboo activated carbon in a multi-layer filtration system. In their study, activated carbon was placed in a glass funnel, and source water flowed from top to bottom under gravity at a controlled rate of 5 ± 2 mL/min, resulting in a 50% reduction in total hardness in water from Chiayi Lantan Lake.
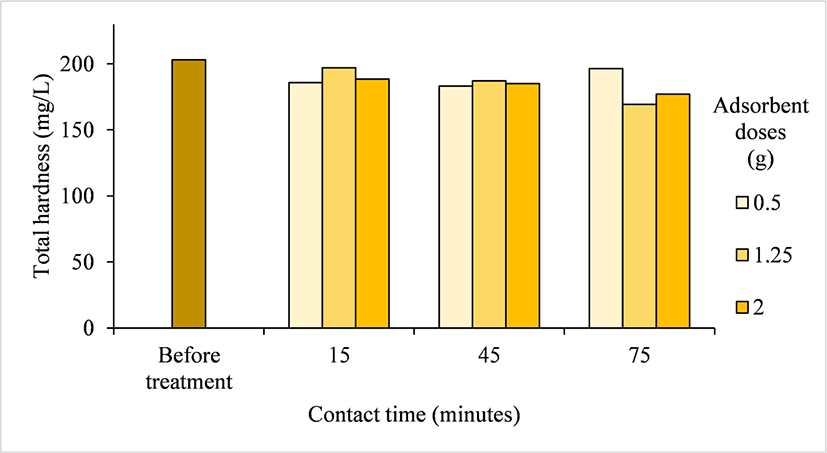
The capacity of activated carbon to reduce total hardness is attributed to its porous surface; the higher the pore capacity or surface area, the greater the ability to reduce the total hardness in water (Adewuyi and Olabanji, 2022; Lin et al., 2017; Nurhayati et al., 2021).
4. CONCLUSIONS
Based on our findings, we concluded that the interaction between the adsorbent dose and contact time of activated carbon and well water significantly affects the Mn concentration. The adsorbent dose also significantly influences the pH and turbidity of well water. The optimal treatment parameters for the Pelemsewu well water with Ampel bamboo waste-derived activated carbon was a 2 g/L adsorbent dose and 75-min contact time, which reduced Fe concentration by 87.50% (from 0.56–0.07 mg/L), Mn concentration by 49.91% (from 0.81–0.41 mg/L), color content by 95% (from 20–1 TCU), turbidity by 98.90% (from 100–1.10 NTU), and total hardness concentration by 12.75% (from 202.99–177.11 mg/L). However, the treatment only increased pH slightly by 18.67% (from 7.5–8.9). These results suggest that Ampel bamboo waste-derived activated carbon is a promising material for improving well water quality, especially in the Bantul District, Special Province of Yogyakarta, Indonesia. After treatment, the Fe concentration in the well water met the Indonesian standards for safe drinking water outlined by the Ministry of Health Regulation No. 2/2023. In conclusion, activated carbon derived from Ampel Bamboo waste is an environmentally friendly and biodegradable material that can enhance the quality of well water for community use. For further development, future research should focus on designing a simple and practical water filtration system based on this material.
