1. INTRODUCTION
Sustainable development and environmental protection have become global imperatives, driving increased interest in eco-friendly materials across industries. Cork has attracted attention as a low-carbon, renewable, and biodegradable biomaterial with favorable durability and water resistance, and its applications have expanded beyond wine stoppers to construction, automotive components, and acoustic insulation (Abera, 2024; Barrigón Morillas et al., 2021; Bontempi et al., 2021; Danworaphong and Eadkhong, 2025; Gil, 2009; Miranda and Pereira, 2024; Tazmeen and Mir, 2024). These functional attributes arise from cork’s unique cellular structure and chemical composition. In particular, suberin an aliphatic–aromatic biopolyester deposited in cork cell walls plays a central role in cork’s barrier performance and long-term durability (Graça, 2015; Graça and Pereira, 2000; Pereira, 2015; Serra and Geldner, 2022). In addition, cork can be harvested without felling the tree, enabling repeated and renewable production cycles (Gil, 2015).
Most cork research and industrial utilization have historically focused on Quercus suber (Q. suber), which is predominantly cultivated in Southern Europe (Marques et al., 2016; Pereira, 2013). In contrast, Quercus variabilis, a cork-forming oak native to East Asia, has received comparatively limited attention. Importantly, comparative studies that disentangle species effects from geographic (provenance) effects on cork chemistry remain scarce, particularly with respect to suberin-related biopolymeric structures and associated chemical fingerprints (Bento et al., 2001; Duarte and Bordado, 2015; Jeong et al., 1988; Miranda et al., 2013; Pereira, 1988). Establishing an origin-aware chemical baseline is therefore essential for material selection and quality control.
In the Republic of Korea (ROK), increasing demand for sustainable construction and interior materials has stimulated interest in cork-based products; however, most cork products are imported and their quality can vary depending on production region, potentially affecting end-use performance (Prades et al., 2012). Moreover, the mountainous terrain of the ROK makes large-scale harvesting of oak cork challenging, increasing the practical value of analytical approaches that can identify materials using small sample sizes (Páscoa et al., 2024). Recent work has also demonstrated that spectroscopy-based screening on bark-related surfaces coupled with chemometric modeling can enable rapid property prediction in lignocellulosic materials (Agustiningrum et al., 2025). We previously conducted a bark-level screening study on six native ROK oak species; based on those results, Q. variabilis was identified as a promising cork-forming species for further investigation (Kim et al., 2025).
Accordingly, the objective of this study is to compare cork derived from Q. suber (Portugal and Italy) and Q. variabilis (ROK and China) to clarify how species and provenance jointly shape cork chemistry. We quantify major structural components (suberin and lignin) and characterize compositional features using Fourier-transform infrared (FT-IR) spectroscopy and pyrolysis–gas chromatography/mass spectrometry (Py-GC/MS). By providing a systematic cross-origin comparison, this work aims to (i) establish a chemical basis for origin-aware material selection and (ii) support the potential utilization of Q. variabilis as a sustainable cork resource in East Asia.
2. MATERIALS and METHODS
Cork materials from two Q. variabilis provenances (China and ROK) and Q. suber provenances (Portugal and Italy) were used in this study. All samples were milled and sieved to obtain 40–80 mesh powder for subsequent analyses. The cork samples from China, Portugal and Italy were supplied as commercial reproduction cork by FC Korea Land (Seoul, Korea) and were ground using a continuous milling system. The Q. variabilis from ROK cork was obtained as virgin cork from the Kangwon National University Experimental Forest and processed in the same manner.
The samples differed in pre-processing history and tissue composition as provided by the supplier. The Portuguese cork was obtained after steam–thermal pressure pre-treatment, whereas the Chinese sample was supplied as cork including bark tissue. In contrast, the Italian cork and the ROK Q. variabilis cork were supplied without pre-treatment and did not include bark tissue. Because the Portuguese, Italian, and Chinese materials were supplied as commercial reproduction cork, the age of the source trees was not available from the supplier; the age information for the ROK virgin cork was also not determined in this study.
The chemical composition of cork, including ash, solvent extractives, suberin, lignin, and polysaccharides, was determined according to the procedures described by Kim et al. (2025) and standard methods for lignocellulosic biomass. Moisture content was determined by the oven-dry method, and all chemical composition data were calculated on an oven-dry weight basis.
Ash and solvent extractives were measured on powdered cork (approximately 2 g) by muffle-furnace combustion and Soxhlet extraction with dichloromethane, ethanol, and hot water following TAPPI T 204 cm-17 (TAPPI, 2017). Sequential solvent extraction was conducted with dichloromethane, ethanol, and hot water at 60°C, 80°C, and 100°C, respectively, for 8 h each, and the extractive-free (EF) residue was designated as EF.
Suberin content was quantified by alkaline methanolysis of EF cork using a 3% sodium methoxide solution in methanol, following the procedure of Graça and Pereira (1997) as applied in our previous work (Kim et al., 2025). Briefly, the EF sample (1.5 g) was reacted in 100 mL (w/v) of 3% NaOCH3 methanol solution for 3 h, filtered, and then reacted again in 100 mL of methanol for 30 min. The methanolysis-derived extract was neutralized by slowly adding it into 1% acetic acid under gentle stirring for 1 h, washed with distilled water until pH 6.5–7, and dried at 60°C for 24 h; this dried fraction was designated as the suberin-rich fraction (SB). The remaining solid residue was designated as the suberin-free residue (SF).
Acid-insoluble (Klason) lignin and acid-soluble lignin were obtained by two-step sulfuric acid hydrolysis of EF samples, where the dried residue after filtration was used to determine Klason lignin and the filtrate absorbance at 280 nm was used to calculate acid-soluble lignin. The same hydrolysates were analyzed by HPLC equipped with an Aminex 87H column and refractive index detection to quantify glucose and xylose, which were used to estimate the polysaccharide content. All compositional determinations were performed using three independent samples per provenance (n = 3).
After the extraction and suberin isolation steps, the color of the SB was measured to compare color differences among the samples. Color was evaluated in the CIELAB color space (L*, a*, b*) using a colorimeter (CR-10 Plus, Konica Minolta, Osaka, Japan). The L* value represents lightness (100 = white; 0 = black), whereas a* and b* represent chromaticity (+a* red/–a* green; +b* yellow/–b* blue). The L*, a*, and b* values were determined following the general CIELAB methodology reported in related studies (Costa et al., 2019; Lee et al., 2023; Park et al., 2022, 2023). Suberin-rich samples were measured in powder form by placing the powder to a thickness of approximately 5 mm in the sample holder and leveling the surface prior to measurement. For each independent sample, color was recorded at three different points (three readings per sample), and three independent samples per provenance were analyzed (n = 3).
Instrument calibration using a white standard tile was not performed in this study, and the results are therefore presented as comparative values obtained under consistent measurement settings.
Attenuated total reflectance Fourier transform infrared spectroscopy (ATR-FTIR) analysis was performed using a Nicolet iS5 spectrometer (Thermo Fisher Scientific, Waltham, MA, USA) to identify the functional groups in each suberin sample. The FT-IR spectra were collected over a range of 500–4,000 cm–1 with 64 scans. Particular emphasis was placed on the FT-IR spectral regions of 2,924–3,000 cm–1, 2,850–2,940 cm–1, 1,730–1,738 cm–1, and 700–1,000 cm–1. FT-IR analyses were performed using three independent samples per provenance (n = 3).
Py-GC/MS analysis was conducted using a pyrolyzer (Pyroprobe 5000, CDS Analytical, Oxford, PA, USA) coupled to a gas chromatograph (7890B, Agilent Technologies, Santa Clara, CA, USA) and a mass spectrometer (5977A, Agilent Technologies) equipped with an HP-5MS column. Approximately 0.7 mg of each sample was placed in a quartz tube, and fluoranthene was used as an internal standard.
Nitrogen was used as the carrier gas at a constant flow rate of 1.0 mL min–1. The GC oven was programmed to hold at 50°C for 5 min, ramp at 3°C min–1 to 280°C, and hold for 10 min. The injector temperature was set to 250°C, and the injector was operated in split mode with a split ratio of 1:15. Pyrolysis products were identified qualitatively using the NIST mass spectral library.
Statistical analyses were conducted to evaluate differences among provenances and species following solvent extraction and methanolysis. One-way analysis of variance (ANOVA) was performed at a significance level of p < 0.05, and Tukey’s post hoc test was applied for multiple comparisons when significant differences were detected. For Py-GC/MS data, the Kruskal–Wallis H test was employed to assess significant differences in chemical composition among groups, and hierarchical clustering using Ward’s method was applied to classify samples based on chemical composition profiles. All statistical analyses were conducted using IBM SPSS Statistics (Version 29.0, IBM, Armonk, NY, USA). Prior to applying parametric tests, data normality was assessed using the Shapiro–Wilk test; if data did not meet normality assumptions, appropriate non-parametric tests were conducted. Unless otherwise stated, all analyses were performed using three independent samples per provenance (n = 3).
3. RESULTS and DISCUSSION
The chemical composition of cork from Q. variabilis (ROK, China) and Q. suber (Portugal, Italy) is summarized in Table 1.
Overall, both species and provenances differed markedly in the relative amounts of extractives, suberin, lignin, and polysaccharides, indicating pronounced species- and origin-dependent variability in cork structure and, consequently, its potential functional behavior.
As shown in Table 1, Portuguese Q. suber showed the highest extractives content (13.6%), which was significantly greater than that of the Q. variabilis samples (p < 0.05). In contrast, Italian Q. suber had a lower extractives content (9.68%), comparable to that of Q. variabilis, highlighting that differences occur not only between species but also among provenances. In all samples, ethanol-soluble extractives were more abundant than dichloromethane- and water-soluble fractions, suggesting that relatively polar compounds constitute a major portion of cork extractives in these materials. Because lipophilic constituents can be informative for wood/cork tissue chemistry and formation-related processes, the DCM-soluble fraction may provide additional compositional cues across provenances (Arisandi et al., 2024).
Suberin, the main structural component of cork, varied significantly with both species and provenance. Q. variabilis corks contained 44%–50% suberin, with the ROK provenance showing the highest value (50.19%). Among Q. suber, the Portuguese sample had the lowest suberin content (30.09%), whereas the Italian sample reached 47.63%, similar to the levels observed in Q. variabilis. These patterns suggest that suberin accumulation is shaped not only by species but also by provenance-associated factors (e.g., growth environment and/or source-related variability of commercial materials). The relatively high suberin contents in Q. variabilis and Italian Q. suber indicate potential suitability for applications where barrier-related behavior and hydrophobic character are advantageous, consistent with previous reports on high-quality cork (Saiz-Fernández et al., 2022).
In contrast, total lignin content tended to be higher in Q. suber than in Q. variabilis. Portuguese Q. suber exhibited the highest lignin content (24.90%), approximately 5.5 percentage points higher than that of Italian Q. suber (19.42%). The Q. variabilis samples from ROK and China showed intermediate lignin levels (18.65% and 21.80%, respectively). Because lignin contributes to structural rigidity and resistance to deformation, higher lignin contents are often associated with increased stiffness and durability of cork-based materials (Leite and Pereira, 2017), consistent with recent studies on cork composite boards exhibiting improved mechanical strength through lignin–fiber interactions (Cha et al., 2022).
Accordingly, the composition of Portuguese Q. suber with lower suberin but higher lignin may support comparatively higher rigidity, whereas suberin-rich corks (e.g., Q. variabilis and Italian Q. suber) may favor barrier-related and compliance-related performance.
Notably, these property implications remain hypothesis-driven in the present study because direct mechanical testing was beyond the scope of this work.
Conversely, the lower lignin contents in Q. variabilis corks may imply lower stiffness than Q. suber, while their higher suberin levels may contribute to flexibility and water repellence.
The differences in lignin content between ROK and Chinese Q. variabilis also suggest that provenance-associated conditions may modulate lignin accumulation and, consequently, potential performance.
The polysaccharide content displayed significant regional variations, even within the same species. In Q. variabilis, the Chinese sample contained 20.38% polysaccharides, while the ROK sample had a lower content of 17.11%. In Q. suber, the Portuguese sample exhibited the highest polysaccharide content at 27.46%, whereas the Italian sample showed a comparatively lower content of 20.12%. These results indicate that carbohydrate-associated fractions are influenced by provenance as well as species. While direct performance testing was beyond the scope of this study, such compositional differences may affect surface interactions and impregnation behavior in cork-based materials, as suggested by studies on chemically modified or delignified wood surfaces (Park et al., 2023).
In the analysis of monosaccharides, glucose emerged as the most abundant sugar across all samples. Among the Q. suber samples, the Portuguese specimen exhibited the highest glucose content at 13.20%, followed by the Italian sample at 9.77%. In contrast, Q. variabilis displayed lower glucose levels, with the ROK sample containing 7.36% and the Chinese sample 8.13%. These results indicate regional variations in carbohydrate accumulation. Other monosaccharides, such as galactose, xylose, and arabinose, were present in smaller quantities, with their concentrations differing across various regions. Because carbohydrates contribute to the cell-wall matrix and can influence porosity and structural cohesion, provenance-dependent carbohydrate variation may also affect processing behavior and end-use performance of cork-based materials, although this relationship requires dedicated physical testing for confirmation.
Phenolic compounds and conjugated structures can contribute to color formation in lignocellulosic materials (Chen et al., 2012; Park et al., 2022; Sundqvist and Morén, 2002; Vinha Zanuncio et al., 2015; Wei et al., 2017). In cork, lignin and phenolic extractives are generally associated with darker and more yellow–brown hues, while SBs typically appear lighter due to their aliphatic polyester nature and comparatively lower aromatic density.
Fig. 1 illustrates the visual appearance of EF, SB, and SF cork powders from the four provenances. EF and SB powders appear beige to yellow, while SF residues are visibly darker and more brownish. These impressions are consistent with the CIELAB color values of SB and SF (Table 2). For all provenances, SB fractions show higher L* values (38.0–56.8) than the corresponding SF fractions (25.0–29.4), confirming that SB is systematically lighter than SF. Likewise, b* values are consistently higher in SB (10.9–33.0) than in SF (14.9–27.6), indicating a more pronounced yellow component in the SB. The a* coordinate does not exhibit a uniform trend among provenances, suggesting that redness is less systematically affected than lightness and yellowness.

| Q. variabilis | ||||||
|---|---|---|---|---|---|---|
| Republic of Korea | China | |||||
| L* | a* | b* | L* | a* | b* | |
| SB | 38.0 | 11.5 | 10.9 | 56.8 | 16.5 | 28.5 |
| SF | 27.9 | 19.9 | 27.6 | 28.7 | 16.0 | 21.5 |
| Q. suber | ||||||
|---|---|---|---|---|---|---|
| Portugal | Italy | |||||
| L* | a* | b* | L* | a* | b* | |
| SB | 41.1 | 17.9 | 31.5 | 40.2 | 19.1 | 33.0 |
| SF | 29.4 | 16.1 | 19.3 | 25.0 | 13.8 | 14.9 |
Compared with our previous outer-bark screening, the present cork fractions display distinct yellow-to-brown hues, supporting the view that differences in the balance of suberin versus lignin/phenolic structures contribute to the observed color variability among species and provenances. Based on these visual and colorimetric trends, FT-IR and Py-GC/MS analyses of SB and SF were carried out to clarify how these differences relate to the underlying chemical structures of cork.
ATR-FTIR spectroscopy was used to confirm the presence of typical suberin structures and to compare the chemical features of SB from Q. variabilis (ROK, China) and Q. suber (Portugal, Italy). Spectra were recorded in the range 4,000–500 cm–1, and the main bands are shown in Fig. 2.
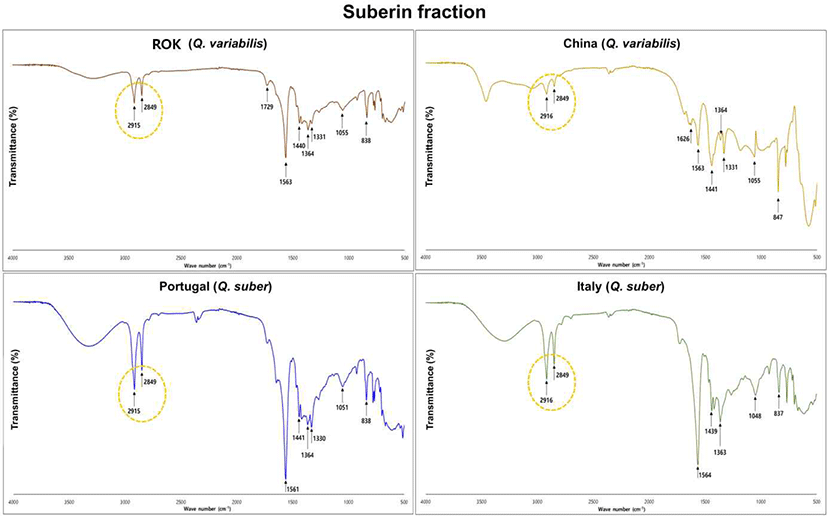
All four spectra display strong absorption bands at 2,915–2,916 and 2,849 cm–1, assigned to asymmetric and symmetric CH2 stretching vibrations of long-chain aliphatic groups. A distinct band at approximately 1,729 cm–1 is observed in every sample and can be attributed to ester C = O stretching in the aliphatic polyester network of suberin. Together, these features confirm that the isolated SB fractions retain the characteristic long-chain ester structure of cork suberin.
In the 1,600–1,500 cm–1 region, bands near 1,561–1,564 cm–1 indicate contributions from aromatic skeletal vibrations associated with lignin- or phenolic-related moieties within the SB fraction.
Notably, the Chinese Q. variabilis suberin shows an additional band at 1,626 cm–1 that is not evident in the other spectra, suggesting a provenance-dependent difference in aromatic/conjugated environments.
Below 1,500 cm–1, bands near 1,440 cm–1 and 1,363–1,364 cm–1 correspond to CH2 bending and CH3 deformation modes, respectively. In the 1,100–1,000 cm–1 region, bands at 1,055–1,048 cm–1 are assigned to C–O stretching in ester and/or polysaccharide structures, and peaks at 838–847 cm–1 are consistent with out-of-plane C–H deformation in substituted aromatics or alkene = C–H bending. These bands are present in all spectra, although their relative intensities differ slightly between Q. variabilis and Q. suber samples.
In similar studies analyzing the cork properties of the ROK Q. variabilis species, comparable spectral patterns were observed; however, the present study differs in that only suberin was extracted and analyzed (Prasetia et al., 2024). Overall, the FT-IR spectra verify the coexistence of long-chain aliphatic esters and aromatic/phenolic groups in all SB fractions, while minor shifts and intensity differences among provenances indicate subtle, species- and region-dependent variations in suberin chemistry.
Py-GC/MS is widely used to characterize lignocellulosic materials and provides semi-quantitative profiles of thermally released fragments from suberin and lignin.
In this study, it was applied to the SB fractions extracted from Q. variabilis and Q. suber to examine species- and provenance-dependent differences in aliphatic and aromatic composition among provenances.
Fig. 3 shows the Py-GC/MS pyrograms of suberin from the four provenances. All samples exhibit a series of intense peaks corresponding to long-chain aliphatic products, together with smaller peaks assigned to lignin-derived aromatics. In agreement with Lourenço et al. (2020), the aliphatic products detected here span approximately C6–C20, including representative 1-alkenes, n-alkanes and related carbonyl compounds (e.g., 2-octene, octane, 1-decene, decane, 1-dodecene, dodecane, 1-tridecene, tridecane, 1-hexadecene, hexadecane, 1-heptadecene, heptadecane, 1-octadecene, octadecane, 1-nonadecene and nonadecane).
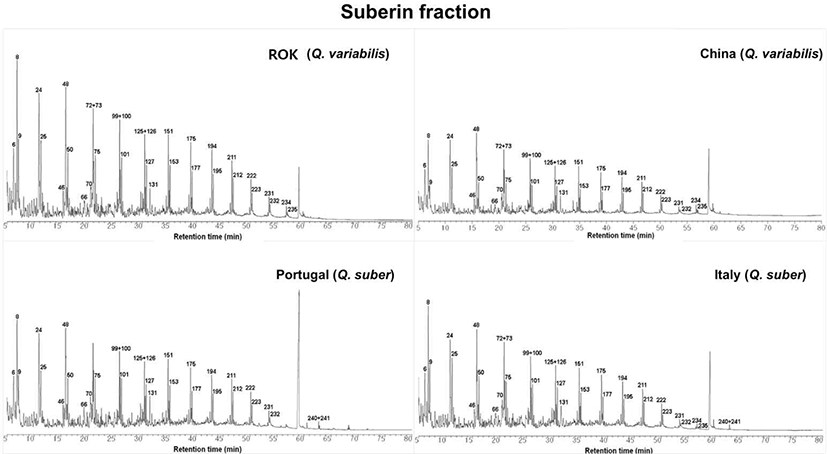
Across all pyrograms, the aliphatic peaks clearly dominate over the aromatic peaks, confirming the aliphatic polyester nature of cork suberin. Nevertheless, small but distinct phenolic and guaiacyl/syringyl-type peaks are present, indicating that a limited amount of lignin-related structures is associated with the suberin fraction. Visual inspection of Fig. 3 suggests that Italian Q. suber has particularly strong α-alkene and alkadiene peaks, whereas Chinese Q. variabilis shows a more pronounced alkane pattern, foreshadowing the quantitative trends summarized in Table 3.
Data represent median values (mg g–1) for various compound types, including alkanes, alkenes, alkadienes, and aromatic compounds.
The semi-quantitative composition of the SB fractions is summarized in Table 3. Total aliphatic products were highest in Italian Q. suber (161.1 mg g–1), followed by Chinese Q. variabilis (103.42 mg g–1) and ROK Q. variabilis (98.62 mg g–1), while Portuguese Q. suber showed the lowest aliphatic content (88.89 mg g–1).
Within the aliphatic group, Chinese Q. variabilis exhibited the highest alkane content (56.71 mg g–1), whereas Italian Q. suber was distinguished by very high amounts of other alkenes (112.29 mg g–1) and alkadienes (19.26 mg g–1). These patterns indicate that Italian Q. suber SB is strongly enriched in unsaturated aliphatic products, while Q. variabilis particularly the Chinese provenance shows higher contributions from saturated alkanes.
Such differences may influence oxidative reactivity and processing behavior; however, direct validation requires targeted experiments beyond the present scope.
Aromatic products were also strongly provenance-dependent. Chinese Q. variabilis showed the highest aromatic content (63.34 mg g–1), followed by ROK Q. variabilis (46.78 mg g–1) and Italian Q. suber (43.34 mg g–1), whereas Portuguese Q. suber had the lowest value (18.56 mg g–1).
In all cases, non-phenolic aromatics accounted for the majority of aromatic products, but phenolic compounds were also significant, ranging from 9.26 mg g–1 in Portuguese Q. suber to 18.09 mg g–1 in Italian Q. suber.
Because phenolic products are often linked to antioxidant-related chemistry, elevated phenolic contributions may be relevant to stability and aging behavior, although this implication remains indirect without dedicated functional testing (Arisandi et al., 2025; Putri et al., 2025). Lignin-derived moieties present in the SB fractions are detailed in Table 4. Their absolute amounts (≈3–5 mg g–1) are much lower than the total aliphatics, consistent with a minor lignin contribution to suberin. The syringyl-to-guaiacyl (S/G) ratio in SB is low for all samples (0.09–0.32), and the H:G:S ratios indicate that H- and G-type units dominate over S units. Italian Q. suber shows a slightly more balanced H:G ratio (1:1.1:0.1), whereas the other provenances display more G-enriched patterns.
Following alkaline methanolysis, the SFs represent the cork matrix depleted in aliphatic suberin and enriched in lignin and carbohydrate-derived structures.
Py-GC/MS analysis of SF therefore provides complementary information on the lignin-dominated aromatic network remaining after suberin removal.
Fig. 4 shows the pyrograms of SF from the four provenances. Compared with SB, the SF pyrograms display a pronounced decrease in aliphatic peaks and a strong relative enrichment in lignin-derived aromatics and phenolics. Major peaks are assigned to phenol, cresols, xylenols, guaiacol and syringol derivatives (e.g., phenol, 4-ethyl-; 2,6-xylenol; 2,5-xylenol; 4-ethyl-guaiacol; eugenol; vanillin; 2-methoxy-4-vinylphenol), indicating that the SF residues are dominated by a lignin-rich aromatic domain. This qualitative shift from aliphatic to aromatic dominance reflects the removal of the aliphatic polyester suberin and the exposure of the underlying lignin framework.
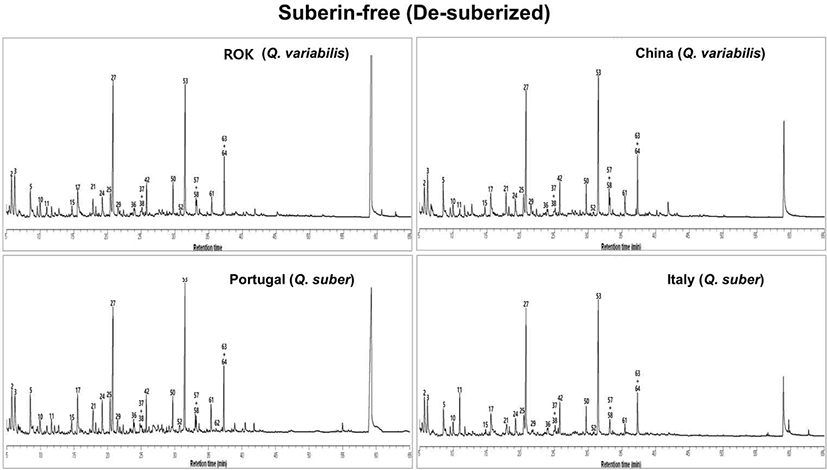
The lignin-derived products in SF are summarized in Table 5. Total lignin moieties in SF are about five- to seven-fold higher than in the SB fractions, confirming that most lignin remains in the residue after suberin extraction. The SF residues of Q. variabilis (ROK and China) and Portuguese Q. suber show similar total lignin levels (20.97–22.19 mg g–1), whereas the Italian Q. suber residue contains substantially less lignin (6.63 mg g–1). The distribution of H, G and S units exhibits clear species- and provenance-dependent patterns. In Q. variabilis SF, G-type units dominate, leading to low S/G ratios of 0.10–0.12 and H:G:S ratios of 1:2.7:0.3 (ROK) and 1:3.0:0.3 (China), indicative of strongly G-enriched lignin networks. Portuguese Q. suber shows a slightly less G-rich pattern (H:G:S = 1:1.8:0.2) but still low S/G (0.12). In contrast, Italian Q. suber SF has a markedly higher S/G ratio of 0.60 and an H:G:S ratio of 1:0.8:0.5, reflecting a more S-rich and less G-dominated lignin structure.
Because lignin unit composition can influence crosslinking density, rigidity, and degradation behavior, these differences may contribute to provenance-dependent performance differences in cork-based materials; however, direct mechanical and durability testing will be needed to confirm these functional implications.
4. CONCLUSIONS
The present results demonstrate that species and geographic origin jointly shape cork chemistry at both bulk and molecular levels, providing a provenance-aware chemical baseline for cork from Q. variabilis and Q. suber.
-
Cork from four provenances (ROK, China, Portugal, and Italy) showed clear differences in extractives, suberin, lignin, and polysaccharides. Q. variabilis (ROK and China) and Italian Q. suber were comparatively suberin-rich, whereas Portuguese Q. suber had lower suberin but higher lignin and polysaccharides.
-
Color measurements and FT-IR spectra of the SBs confirmed long-chain aliphatic ester structures in all samples, with minor aromatic/phenolic features. SB powders were consistently lighter and more yellow than SFs, consistent with higher L and b values and stronger aliphatic ester-related FT-IR bands.
-
The fractionation procedure effectively separated a SB while leaving lignin- and carbohydrate-enriched residues. Py-GC/MS further differentiated species and provenances at the molecular level.
-
SB fractions were dominated by C6–C20 aliphatic products. Italian Q. suber was enriched in unsaturated aliphatics (α-alkenes and alkadienes), whereas Chinese Q. variabilis showed higher contributions from saturated alkanes and a higher aromatic yield.
-
Lignin-derived products were minor in SB and showed low S/G ratios. In contrast, SF residues contained approximately five- to seven-fold higher lignin-derived products and distinct H–G–S distributions: Q. variabilis SF was strongly G-dominated (low S/G), while Italian Q. suber SF showed a higher S/G ratio and a relatively more S-rich lignin pattern.
These findings may support origin-aware classification and material selection. However, application-related implications remain hypothesis-driven without direct mechanical, barrier, and weathering tests; future work should therefore integrate targeted performance evaluations with the chemical fingerprints established here and explore additional valorization routes for aromatic-rich lignin-derived constituents.








